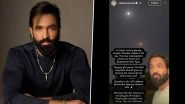
The Subject Experts Committee (SEC) of the Drug Controller General of India (DGCI) granted "in principle" approval to #BharatBiotech to conduct "Phase III superiority study & Phase III booster dose study" trials for its intranasal #CovidVaccine. pic.twitter.com/gAF39hn3KU— IANS Tweets (@ians_india) January 5, 2022
(SocialLY brings you all the latest breaking news, fact checks and information from social media world, including Twitter (X), Instagram and Youtube. The above post contains publicly available embedded media, directly from the user's social media account and the views appearing in the social media post do not reflect the opinions of LatestLY.)












 Quickly
Quickly


